As an epidemiologist and mother, I never imagined we would be weighing the risk of measles in America again.
Published March 10, 2026
By Syra Madad, D.H.Sc., M.Sc., MCP, CHEP
My fourth child will turn 1 soon. She is healthy, curious and full of the spark that makes early childhood feel so wondrous. But unlike my older children, she entered infancy at a moment when measles, a disease the United States eliminated in 2000, feels uncomfortably close again.
What makes this especially painful is that measles is not some mysterious new threat. It is a vaccine-preventable disease. The United States declared measles eliminated in 2000, meaning endemic transmission had been stopped. We still officially retain that status. But elimination does not mean the virus is gone forever. It means cases are usually imported from elsewhere and do not sustain continuous transmission here for 12 months or more. That protection depends on high vaccination coverage and fast public health response. Both are now under strain.
Flirting with Disaster
When my daughter was 6 months old, I made the decision to give her an early dose of the measles, mumps and rubella vaccine. Now, as she approaches her first birthday, I am profoundly relieved that she will soon be eligible for the routine dose that counts as the first in her standard series. That relief says as much about the state of public health in America as it does about my instincts as a mother.
The numbers are sobering. As of March 6, the Centers for Disease Control and Prevention had recorded 1,281 confirmed measles cases in the United States in 2026. That is just the first three months of the year. For comparison, the country recorded 2,281 confirmed cases in all of 2025 and 285 in all of 2024. In other words, by early March, this year had already seen nearly four times as many measles cases as the entire year before last. CDC says 90% of confirmed U.S. measles cases this year are outbreak-associated, including outbreaks that began in 2025 and are still spilling into 2026.
The stakes are no longer theoretical. A formal review of the United States’ measles elimination status, originally expected this spring, has now been delayed until November. Whatever the reason for the delay, the fact that such a review is even necessary should alarm us. A country that eliminated measles a quarter-century ago should not be flirting with the possibility of losing that hard-earned distinction.
The Unraveling of the Social Contract
A recent modeling study projected that if vaccination rates continue to decline, the United States could see millions of measles cases over the next 25 years and face the return of endemic spread. Another finding was more hopeful: a modest 5 percentage-point increase in MMR vaccination could dramatically reduce future measles burden. Prevention still works, if we choose it.
As an infectious disease epidemiologist, I have spent the last two decades studying emerging outbreaks and public health preparedness. Measles, one of the most contagious viruses known, is not a mild childhood illness. It can cause pneumonia, brain swelling, blindness and death. Even after recovery, children can suffer immune suppression that leaves them more vulnerable to other infections.
For babies like mine, especially under 1 year old, the threat is even more serious. Their immune systems are still developing, and they are too young to be fully protected by the standard two-dose vaccine schedule. Infants carry maternal antibodies that help protect them during the first months of life. But those antibodies fade, leaving babies in a gray zone: no longer protected from measles, but still carrying enough maternal antibodies to reduce how well a vaccine works. That is one reason routine MMR vaccination begins at 12 months in lower-risk settings, even though earlier doses are recommended during travel and some outbreaks. For years, we relied on herd immunity to shield them. That social contract is now unraveling.
Striving for 95%
CDC notes that preventing measles outbreaks generally requires about 95% vaccination coverage. National MMR coverage among kindergartners has fallen from 95.2% in the 2019-2020 school year to 92.5% in 2024-2025, leaving roughly 286,000 kindergartners without documentation of complete MMR vaccination. National averages also hide something even more dangerous: local pockets where coverage is far lower. In Idaho, for example, kindergarten MMR coverage was just 79.5% in the 2024–2025 school year. That is where outbreaks ignite. The overwhelming majority of recent measles cases are in people who are unvaccinated or whose vaccination status is unknown, 92% so far in 2026 and 93% in 2025.
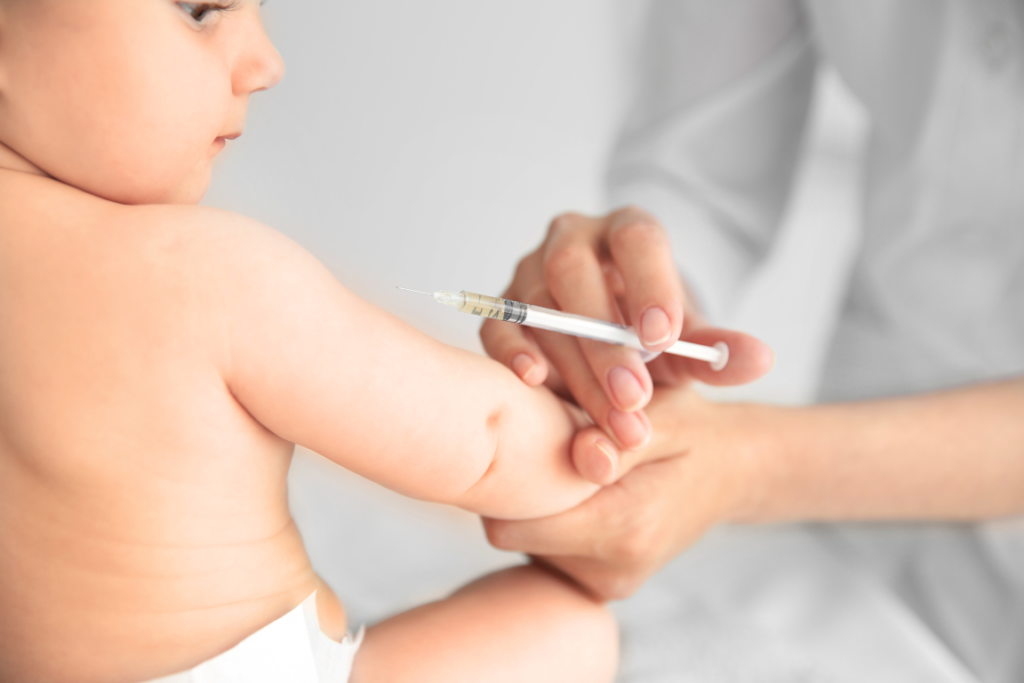
When my daughter was 6 months old, the risk calculus had changed. CDC recommends that infants ages 6 through 11 months receive one dose of MMR before international travel, and public health officials may also recommend early vaccination in outbreak settings. Importantly, that early dose does not replace the routine series. Babies who receive MMR before their first birthday still need another dose at 12 through 15 months and a final dose later in childhood. So as my daughter turns 1, I am not thinking, thankfully, that she is done. I am thinking, thankfully, that she can now begin the part of the schedule that offers more durable protection.
As a mother, I am grateful we had that option. As an epidemiologist, I am troubled that so many families now need to consider it.
Trust, Science, Action
Parents should not have to weigh the risk of measles when planning a trip, boarding a plane or visiting relatives. They should not have to track outbreak maps to decide whether to attend a wedding or take a baby through an airport. This is not an abstract epidemiologic trend. It is the kind of threat that shapes how a parent thinks about a waiting room, an airport gate, a birthday gathering or a family trip.
I am often asked what we can do to stop this. The answer is not complicated. It is trust. It is science. And it is action.
We must rebuild public confidence in vaccines through transparent communication, engagement with local leaders and listening to parents’ concerns without judgment. That is far more difficult when some of the country’s most powerful health officials have spent years undermining vaccine trust. Health Secretary Robert F. Kennedy Jr. now says the MMR vaccine is the most effective way to prevent measles, but his broader vaccine agenda has sent the opposite signal: more doubt, more confusion and less confidence in the routine immunizations that protect children.
We must invest in the public health infrastructure that quietly protects us every day. And we must stop treating routine childhood vaccination as a political battleground.
My daughter is lucky. She got an early dose when she needed it, and now she is old enough to begin the routine schedule that offers stronger, longer-lasting protection. But babies should not need backup plans against a disease the United States eliminated a quarter-century ago.
Measles belongs in history, not in American childhood again.