Exploring vaccine treatments for Dengue, Meningococcal, and Pneumococcal, all of which pose a major public challenge in areas like Southeast Asia, Africa, and Latin America.
Published August 11, 2009
By Theresa M. Wizemann

Despite the availability of life-saving vaccines, communicable diseases remain a major public health problem around the world. There is a gap between when vaccines and technologies become available in industrialized countries, and when they are implemented in countries that need them most, where the disease burden is the greatest. This problem persists because of challenges in research and development, prioritization, regulation, funding, infrastructure development, implementation, and surveillance and monitoring.
A May 20, 2009, symposium hosted by The New York Academy of Sciences (the Academy) looked closely at these issues, focusing on meningococcal and pneumococcal diseases, dengue, and dengue hemorrhagic fever. In addition to explaining the state of the art in vaccine development for these diseases, speakers offered several strategies for protecting populations from vaccine-preventable disease: achieve high immunization coverage rapidly and across age groups with the highest disease burden; assess waning immunity; and do not underestimate carriage reservoirs that can reintroduce the organism into the population.
They also discussed the importance of public–private partnerships to ensuring timely, equitable, and sustainable delivery of lifesaving vaccines to developing countries. Some aspects of successful strategies include: regional demand estimates and bulk purchasing, allowing for lowest price negotiation; purchase commitments and annual contracts, allowing manufacturers to plan for production, delivery, and financial investment; and lines of credit to countries, ensuring uninterrupted delivery of vaccines.
Closing the Gap
Despite advances in treatment and prevention, communicable diseases remain a major public health problem in much of the world. Thirty-two percent of all deaths worldwide are due to infectious diseases, and there is a stark inequality in the global disease burden of vaccine-preventable diseases. The developing world, including regions of Southeast Asia, Africa, and Latin America, bear the greatest burden, in part because life-saving vaccines that are part of routine care in industrialized countries still have not been broadly implemented in these regions.
On May 20, 2009, leading scientists from academia, industry, and nonprofit organizations gathered at the Academy to discuss the unique challenges of developing, evaluating, funding, and delivering vaccines for infectious diseases to those that need them most. The symposium, moderated by Albert Ko of Weill-Cornell Medical College, focused on global efforts to prevent meningococcal and pneumococcal diseases, dengue, and dengue hemorrhagic fever.
The challenges of providing these vaccines to the poorest of the world span research and development, prioritization, regulation, funding, infrastructure development, implementation, and surveillance and monitoring. To close the gap, all of these areas must be dealt with concomitantly.
Dengue, Once Nearly Eliminated, Re-emerges with a Vengeance
Dengue hemorrhagic fever is often classified as an emerging infectious disease, Scott Halstead of the Pediatric Dengue Vaccine Initiative (PDVI) said. The often fatal disease has been amplified by factors of the contemporary world. Global population growth, rural-to-urban migration, and deterioration of cities provide breeding space for the dengue vector, the Aedes aegypti mosquito. Jet travel by people unaware they are infected allows the virus to relocate rapidly. In addition, following eradication of A. aegypti from the American tropics in the 1960s in response to yellow fever, the number of vector control experts dwindled and Aedes has reestablished itself in an even wider range than before.
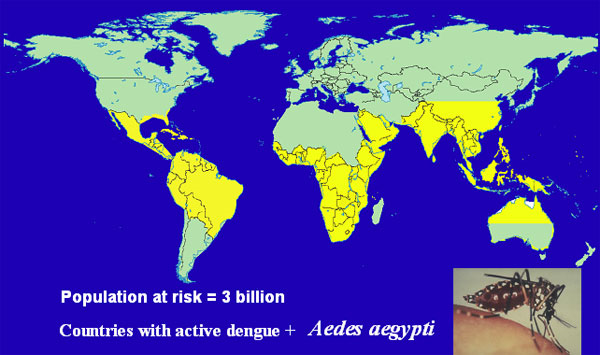
There are four dengue viruses, all endemic in tropical regions of the world. Certain populations, including Africans, appear to have a resistance gene and experience severe disease less frequently. Most observable disease in Africa is in expatriate residents. About half the world’s population, about 3 billion people, live in the tropical regions where the dengue vector thrives. Conservatively, 50–100 million people are infected with dengue each year and about 10% have overt symptoms ranging from mild disease to fatal hemorrhagic fever. The economic impact of dengue illness is considerable.
The majority of severe disease results from secondary infections in those with circulating antibody from a previous infection. Primary infection in infants can also be severe, progressing as if it were a secondary infection due to the presence of maternal antibody. Antibodies interact with dengue virus to form immune complexes that are internalized by monocytic cells, resulting in a complicated phenomenon called intrinsic Antibody Dependent Enhancement (ADE).
In the presence of enhancing antibody (i.e. circulating antibody below protective levels) the number of cells that are infected is greater, and the viral output of each cell is increased. A recent study suggests that while the interferon system is generally effective in controlling infection, in the presence of dengue antibody, immune complexes suppress the interferon system, resulting in more severe disease.
Given the lack of effective vector control and the vast population at risk for contracting dengue, a vaccine is the best hope for controlling the disease. Halstead highlighted five products in development. ChimeriVax-Dengue is a tetravalent dengue vaccine engineered by Sanofi Pasteur by inserting structural genes from the four dengue viruses into an attenuated yellow fever virus. A second tetravalent vaccine being codeveloped by GSK and the Walter Reed Army Institute for Research (WRAIR) is comprised of native whole dengue viruses, attenuated by serial passage.
Two other chimeric tetravalent vaccine candidates are being developed by NIH in collaboration with Johns Hopkins University, and by the CDC in collaboration with Inviragen, based on attenuated dengue 4 and dengue 2 viruses, respectively. Finally, Hawaii Biotech has expressed the four dengue envelope genes in yeast cells. One of the challenges of vaccine development, Halstead said, is targeting the age when maternal antibodies decline sufficiently to allow a live attenuated vaccine to be effective in infants. Achieving herd immunity through mass vaccination is another approach to better protect infants, and to avoid ADE.
The PDVI supports the development of dengue vaccines, preparing field sites for vaccine trials, working to improve and standardize dengue diagnostic tests, and preparing regulatory and manufacturing standards. PDVI is also working to better understand the burden of illness, strengthen surveillance, and upon approval of a vaccine, will advocate for introduction and use, addressing issues of access.
Meningococcus: Preventing Outbreaks and Controlling Endemic Disease
While dengue virus infects only those who come in contact with its mosquito vector, nearly all humans are colonized with Neisseria meningitides at some point. Emil Gotschlich of the Rockefeller University said that about 10% of the population harbors meningococcus asymptomatically in the nasopharynx in winter months, but in schools, prisons, and the military, the carrier rate can be as high as 60%. Since WWII, there has been a relatively low-level endemic pattern of meningococcal meningitis in the U.S., about 2 cases per 100,000 people, with occasional regional outbreaks.
With the essential elimination of Haemophilus influenzae meningitis as a result of vaccination, meningococcus is now the leading cause of bacterial meningitis in U.S. Overseas, epidemic disease is a serious concern. Most prominent is the “meningitis belt” spanning sub-Saharan Africa. There, endemic disease is very common and major epidemics occur at frequent intervals. There is a rapid onset of cases in the winter months, as high as 500/100,000, which halts rapidly when the rainy season starts. This dependence on meteorological conditions is not fully understood.
The meningococcus is covered with a polysaccharide capsule which is required for virulence, and natural immunity is dependent on circulating antibody to the capsule. A relatively limited number of capsular serogroups cause invasive disease, including types A, B, C, Y, W-135, and X. The principle of vaccination with capsular polysaccharide was established by early studies of the pneumococcal vaccine, and trials of first-generation meningococcal polysaccharide vaccines in school-age children and adults established that the efficacy of vaccines to group A and C meningococci was 90%.
However, infants and young children under the age 2, who are at the highest risk of meningococcal disease, do not respond immunologically to polysaccharide antigens. To address this, researchers focused on developing protein conjugate vaccines modeled after the current H. influenzae vaccines. The current meningococcal conjugate vaccine is tetravalent, covering groups A, C, Y, and W-135, and has proven to be highly effective in preventing outbreaks of meningitis, and controlling endemic disease.
Conjugate vaccines are a marked improvement over polysaccharide alone, allowing for vaccination in the first years of life, achieving high levels of antibody at all ages, and effectively suppressing the carrier state. However they do have shortcomings, including the need for boosters to maintain protective levels of antibody. Conjugate vaccines, if properly manufactured, are excellent. What is required, Gotschlich said, is a leadership that devises and promotes programs to match the quality of the vaccine.
The African Meningitis Belt
Despite vaccination in response to epidemics, outbreaks of serogroup A meningococcal disease occur year after year across sub-Saharan Africa, explained Thomas Clark of the Centers for Disease Control and Prevention (CDC). Over 250 million people are at risk across the African meningitis belt, from childhood, well into the fourth decade of life.
The Meningitis Vaccine Project (MVP) was created in 2001 to develop a meningitis vaccine that is effective and affordable for Africa, and ultimately, to eliminate epidemic meningitis as a public health problem in sub-Saharan Africa. A unique consortium of partners is contributing serogroup A polysaccharide, carrier protein, conjugation technology, and manufacturing capabilities. Clinical trials in Africans aged 2–29 years of age have shown that the novel meningococcal A conjugate vaccine, MenAfriVac, is immunologically superior to the existing tetravalent conjugate vaccine, Menactra. Comparable results were observed in children aged 12–23 months.
In the United States, infants under age 1 and adolescents aged 16–19 have the greatest disease burden. Since 2005, CDC has recommended meningitis vaccination at 11–12 years of age, but coverage in adolescents has taken effect more slowly than infant programs, with teen coverage reaching only 30% by 2007.
The U.K. experience is somewhat different. In 1999, in response to the increasing burden of serogroup C disease, the U.K. rapidly implemented immunization of infants, young children, and adolescents, achieving over 80% coverage across all ages by 2001. In 2008, for the first time, there were no deaths from serogroup C meningococcal disease in the U.K. A study of over 10,000 U.K. adolescents showed that vaccination reduced the carriage rate of serogroup C meningococci by two thirds within one year, resulting in herd immunity and a reduction in disease across all age groups, including in those who were not vaccinated.
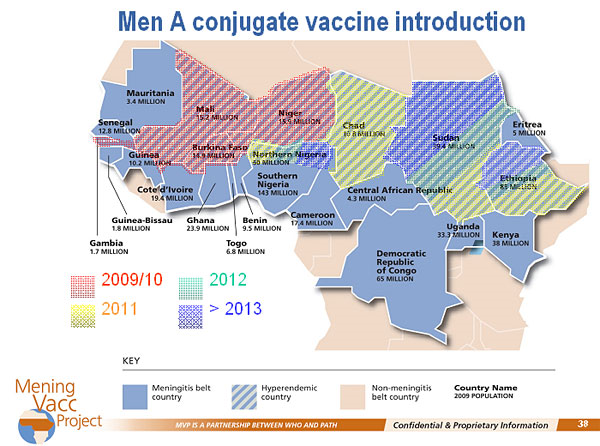
The MVP is modeled on the U.K. experience. Phase 1 is mass vaccination of the population under 30 years of age. Phase 2 includes a catch-up campaign every 5 years in children aged 1 to 5 years, and two infant doses as part of the regular childhood schedule. MenAfriVac has been submitted to the Indian regulatory agency for licensure and a decision is expected in late 2009. Upon approval, Burkina Faso will be the first country to implement vaccination, due in part to the burden of disease, and the ability to conduct surveillance and evaluation. Vaccination will then roll across the meningitis belt.
Even at a cost of 40¢ per dose and about $1 per person to administer, the 12 million doses needed in Burkina Faso are still prohibitively expensive for the country’s ministry of health. Fundraising is ongoing to be able to implement the mass vaccination phase before funding from GAVI is approved. Surveillance will be conducted for coverage levels, vaccine effectiveness, safety, carriage and transmission, outbreaks, and molecular epidemiology, including the emergence of other serogroups.
Pneumococcus: The Number One Cause of Vaccine-preventable Death
Like meningococcus, the primary virulence factor of Streptococcus pneumoniae is the polysaccharide capsule. Pneumococcus is carried asymptomatically in the nasopharynx, and causes clinical disease when it moves to other parts of the body. The most common manifestation is otitis media, infection of the middle ear. More invasive and severe outcomes include pneumonia, bacteremia, and meningitis. Conjugation of pneumococcal polysaccharide to a carrier protein elicits protective levels of functional opsonophagocytic antibody in infants and children.
There are over 90 pneumococcal serotypes, but not all are associated with disease, explained Emilio Emini of Wyeth Pharmaceuticals. The current conjugate vaccine, Prevnar, covers the seven serotypes that are responsible for 80% of invasive disease in children in the U.S.: 4, 9V, 6B, 14,19F, 18C, and 23F. Manufacturing the 7-valent vaccine is extremely complex as it is, in essence, seven separate conjugate vaccines. In clinical trials, the vaccine was shown to be greater than 95% efficacious for invasive disease, and by 5 years after introduction, there was a 98% reduction in invasive pneumococcal disease due to covered serotypes in children. There was also a 76% reduction in disease in unvaccinated adults as a result of herd immunity.
Children in the U.S. have been routinely immunized since 2001; however S. pneumoniae remains the number one cause of vaccine-preventable death in children worldwide. One million infants, and one half million adults, die every year, in large part because the vaccine is just now beginning to be introduced in developing world countries.
The 7-valent vaccine was developed based on serotype prevalence in North America, and the two serotypes that cause epidemic outbreaks of disease in developing world countries, types 1 and 5, are not covered as they are not typically endemic in developed countries. Development of a 9-valent vaccine was undertaken to help address this, and two studies, in South Africa and Gambia, showed the vaccine to be highly effective. The 9-valent vaccine was not fully developed, however, as it became apparent that the epidemiology of the infection was shifting and coverage of more than nine serotypes was needed.
Efforts are now focused on developing a single second-generation vaccine that will be effective worldwide. A new 13-valent vaccine will include serotypes 1 and 5, as well as 3, 6A, 7F, and 19A. All clinical trials have been completed and regulatory approval is now being sought in the U.S. and the EU.
For a vaccine of this complexity, the biggest obstacle to effective delivery to the developing world is manufacturing and cost. The estimated number of pneumococcal vaccine doses required for worldwide use could be as high as 250 million per year. The burden is on biopharmaceutical scientists, Emini said, to improve processes and decrease costs. In addition, WHO requirements must be met before the vaccine is “pre-qualified” (i.e., eligible for purchase by agencies such as the UN that supply vaccines for developing countries). Cold chain requirements also add complexity to delivery and distribution. Emini also noted that political will in the countries in which the vaccine will be made available is required.
In 2009, Wyeth donated enough 7-valent vaccine to immunize every child in Rwanda, which has one of the highest incidences of pneumococcal disease and death. Given the significant unmet medical need, it was decided to begin an immunization program with the current 7-valent vaccine, and transition to the 13-valent as soon as it is licensed.
Breaking the Cycle of High Price, Uncertain Demand, and Limited Supply
Arthur Reingold of the University of Calfornia, Berkeley stressed the dramatic public health impact of the 7-valent pneumococcal vaccine, noting the decrease in disease due to antibiotic-resistant strains, the reduction in noninvasive syndromes such as otitis media, and the decrease in hospitalizations of children due to pneumonia. Children are the primary source of infection for adults, and childhood immunization has led to a significant decline in invasive disease in adults aged 65 and older due to the 7 vaccine serotypes. Remarkably, the indirect herd protection of older children and adults has prevented more disease, and saved more lives, than has direct protection of vaccinated children.
The 7-valent vaccine is administered as three infant doses and a fourth booster dose after age 1 year. But a three-dose schedule could result in a cost savings of 25%, free up global supply, and mean fewer injections for children. Immunogenicity studies have shown that three- and four-dose schedules produce similar antibody levels for most serotypes, and persistence of antibody is similar. The key issue, shown in a case-control study, seems to be that one of the doses must be a booster after the first year of life. Reingold noted that some countries in the EU are already using a three-dose schedule.
The WHO Strategic Advisory Group of Experts (SAGE) recommended introduction of the 7-valent vaccine in the developing world now, switching to the 13-valent when licensed. The challenge now is implementation. There is, in essence, a vicious cycle of high price, uncertain demand, and limited supply, Reingold said.
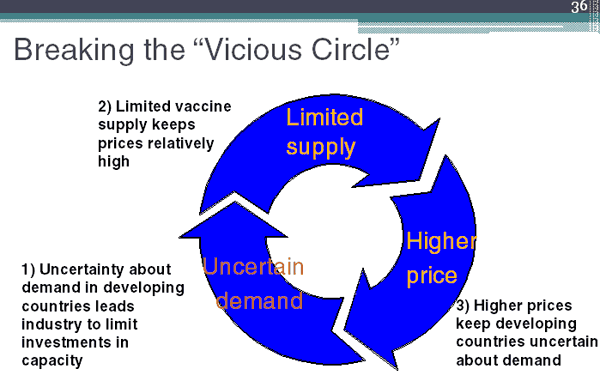
PneumoADIP is working to break this cycle through the development of public–private partnerships. Eligible countries, those with annual per capita income levels of less than $1000, first commit to using the vaccine. Manufacturers, then assured of a market, commit to making and selling the vaccine at a lower price. GAVI and other funders commit to purchasing the vaccine at the guaranteed price for the countries, with a minimal per dose copayment from the country.
This approach, while assisting high-burden countries, does not assist mid-income level countries in the Americas that are not GAVI-eligible, yet cannot afford to pay full price for the vaccine doses they need to give. Some are considering cutting costs by administering fewer doses per child, the health impact of which remains to be seen.
Funding and Delivery: The High Cost of Inaction
Describing his work at the Pan American Health Organization (PAHO), Jon Andrus began by noting that the introduction of vaccines in the Americas (Latin America and the Caribbean) has eliminated or eradicated measles, neonatal tetanus, and polio, and significantly reduced the incidence of diphtheria and pertussis. There is still work to be done, however. In the Americas, two children die every hour of pneumococcal invasive disease. The 7-valent vaccine could prevent at least one of those deaths every hour, Andrus said.
The PAHO experience shows that new vaccines can be successfully introduced and sustained over time. The PAHO Revolving Fund was established 30 years ago to ensure a safe, effective, uninterrupted, sustained supply of vaccines. The Fund uses a bulk purchasing strategy, negotiating best price based on regional demand, rather than per country estimates. A credit system allows for delivery of vaccines in the absence of immediate funds. Countries reimburse PAHO within 60 days after vaccine is delivered, and pay 3% of the price of the vaccine as a service charge to be put into PAHO’s working capital.
This allows for stable and prompt supplies of safe, high-quality, affordable vaccines. For manufacturers, annual contracts facilitate the ability to plan for production, delivery and financial investment. There are reliable demand forecasts, and transparency of relationships. PAHO facilitates “one-stop shopping” for manufacturers, eliminating the need to negotiate individually with 36 member states.
In 2006, the Directing Council of PAHO passed a landmark resolution urging all member states to identify new revenue sources to sustainably finance the introduction of new vaccines against rotavirus, pneumococcus, influenza, and human papilloma virus (HPV). The Revolving Fund policy of single best market price, linked with the power of bulk purchasing, will be critical to accelerated and sustained uptake of these vaccines in participating Latin American countries, Andrus concluded.
If the introduction of pneumococcal and rotavirus vaccines is delayed, almost one million children could die from vaccine-preventable diseases over the next 30 years, and the world would not achieve MDG4, the Millennium Development Goal to reduce child mortality. Ensuring equitable and sustainable introduction of new vaccines into developing countries requires a strategic vision grounded in long-term goals, Andrus emphasized, not short-term fixes.
Moving forward
In summary, to successfully protect populations from vaccine-preventable disease, initiatives should strive to achieve high immunization coverage rapidly, across age groups with the highest disease burden, and should monitor waning immunity. The impact of herd immunity in controlling infectious diseases can be considerable. (i.e. the indirect protection of unvaccinated individuals in a population where a sufficiently high percentage has been vaccinated). However, carriage reservoirs that can reintroduce the organism into the population should not be underestimated. Working as partners, the public and private sectors must develop creative funding approaches to ensure timely, equitable, and sustainable delivery of lifesaving vaccines to developing countries.
Also read: Maternal, Neonatal, and Early Infancy Vaccine Developments